Abstract
Bimekizumab is a humanized monoclonal IgG1 antibody that selectively inhibits IL-17A and IL-17F, two cytokines that play a critical role in psoriasis vulgaris pathogenesis. Randomized clinical trials demonstrating the superiority of bimekizumab compared to placebo and other injectable biologics led to its approval by the United States FDA for moderate-to-severe plaque psoriasis. Due to the often-strict inclusion/exclusion criteria of randomized clinical trials, patient characteristics in real practice may drastically differ. Thus, the authors sought to investigate the use of this medication among real-world patients at their large academic center.
Charts of patients prescribed bimekizumab between October 2023–April 2024 with a diagnosis of moderate-to-severe plaque psoriasis and follow-up within 6 months of medication initiation were included. Results showed that of the 24 patients prescribed bimekizumab, 83.3% (n=20) had concurrent diagnoses of psoriasis and psoriatic arthritis. At bimekizumab initiation, all patients had a baseline body surface area (BSA) affected by psoriasis of ≥10%, with mean BSA being 13.4%. After a mean of 163.5 days of therapy, BSA decreased to 3.8%. Overall, 83.3% of patients experienced a decrease in BSA after initiating bimekizumab, with 37.5% of patients obtaining a BSA of 0% (skin clear). Aligning with clinical trials, this study reinforces bimekizumab as a useful therapeutic option for moderate-to-severe plaque psoriasis. Future real-world research investigating bimekizumab would benefit from increased size and longer follow-up. Despite limitations, this study supports bimekizumab as an appropriate option for treatment of moderate-to-severe plaque psoriasis among patients in real clinical practice.
Key Points
1. Psoriasis affects 2–3% of the population worldwide, with about 30% of these patients developing psoriatic arthritis.2. This retrospective review presents 24 patients with difficult-to-treat psoriasis who had failed prior systemic therapies and were initiated on bimekizumab with positive effects.
3. Bimekizumab can be considered as a next-step therapy for bio-experienced patients, even for patients who have failed another IL-17 inhibitor.
INTRODUCTION
Bimekizumab is a humanized monoclonal IgG1 antibody that selectively inhibits IL-17A and IL-17F, two cytokines that play a critical role in psoriasis vulgaris pathogenesis.1 Randomized clinical trials (RCT) demonstrating the superiority of bimekizumab compared to placebo and other injectable biologics and its clinical safety led to its approval by the United States FDA for moderate-to-severe plaque psoriasis.2 Presently, few studies have investigated the effectiveness of bimekizumab in real clinical practice.3 Due to the often strict inclusion/exclusion criteria of RCTs, patient characteristics in real practice may drastically differ, making real-world studies imperative.4 Herein, the authors describe the use of bimekizumab among 24 patients with difficult-to-treat plaque psoriasis, the majority of whom had previously cycled through multiple systemic therapies.
METHODS
Charts of patients prescribed bimekizumab for psoriasis between October 2023 (time of United States FDA approval) to April 2024 at a large academic center in the United States were retrospectively retrieved and reviewed. For inclusion, a diagnosis of moderate-to-severe plaque psoriasis and follow-up within 6 months of bimekizumab initiation was necessary. The authors defined “difficult-to-treat plaque psoriasis” as that which was refractory to at least two prior systemic therapies.
RESULTS
Of the 24 patients prescribed bimekizumab, 83.3% (n=20) had concurrent diagnoses of psoriasis and psoriatic arthritis; the remaining 16.7% (n=4) of patients had psoriasis only (Table 1). At bimekizumab initiation, all patients had a baseline body surface area (BSA) affected by psoriasis of ≥10%, with mean BSA being 13.4±7.8 (Figure 1). At the start of treatment, the mean age and number of failed systemic therapies was 54.6 years and 4.5 therapies, respectively. All patients (except one) were bio-experienced, and 87.5% of patients had previously cycled through at least two systemic options, with either an inadequate response or secondary loss of efficacy to each.
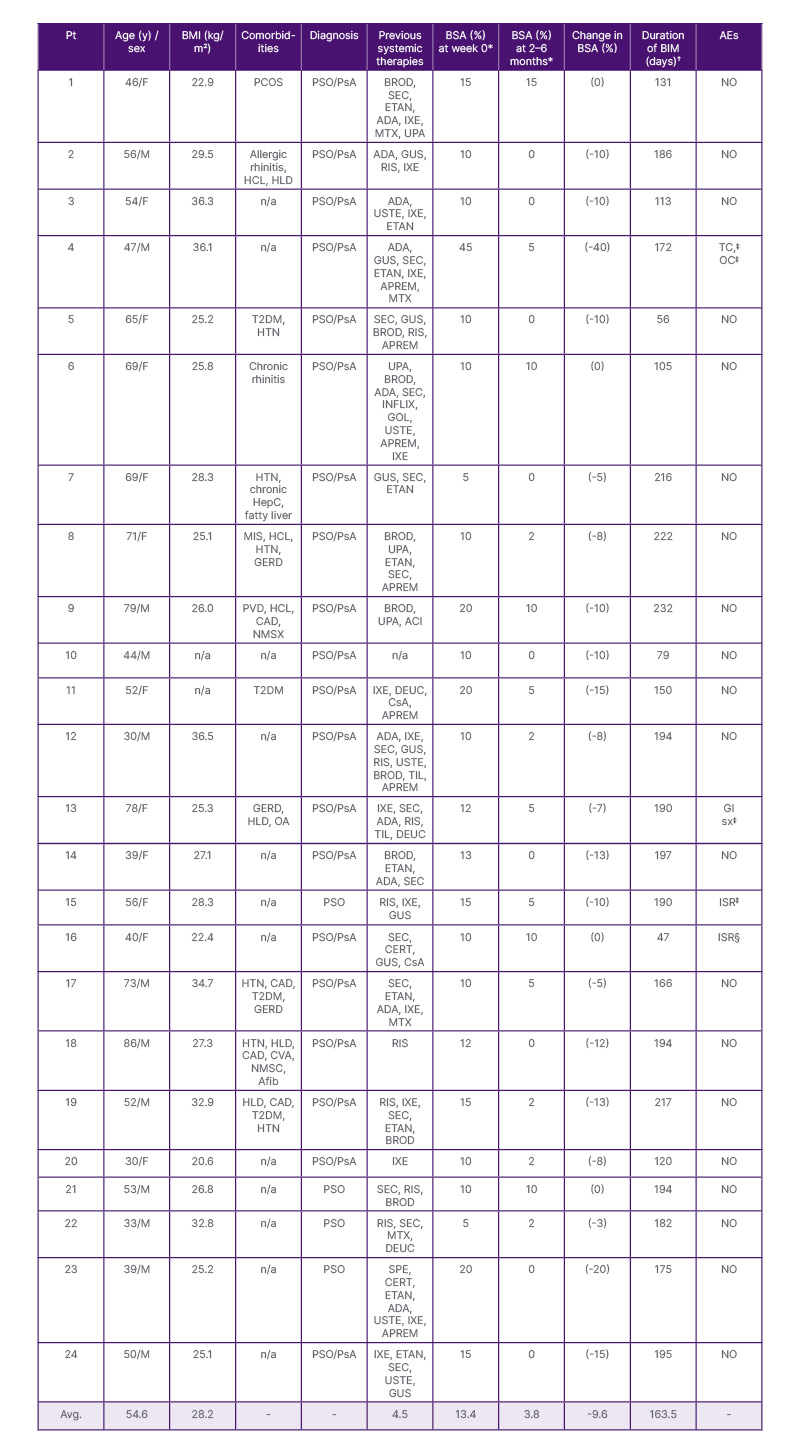
Table 1: Real-world use of bimekizumab in 24 patients with difficult-to-treat plaque psoriasis.
ACI: acitretin; ADA: adalimumab; AE: adverse event; Afib: atrial fibrillation; APREM: apremilast; Avg: average; BIM: bimekizumab; BROD: brodalumab; BSA: body surface area; CAD: coronary artery disease; CERT: certrolizumab pegol; CsA: cyclosporine; CVA: cerebral vascular accident; DEUC: deucravacitinib; ETAN: etanercept; GERD: gastroesophageal reflux disorder; GI: gastrointestinal; GOL: golimumab; GUS: guselkumab; HCL: hypercholesterolemia; HDL: hyperlipidemia; HepC: hepatitis C; HTN: hypertension; INFLIX: infliximab; ISR: injection site reaction;IXE: ixekizumab; MIS: melanoma in situ; MTX: methotrexate; NMSC: non-melanoma skin cancer; OA: osteoarthritis; OC: oral candidiasis; PCOS: polycystic ovarian syndrome; PsA: psoriatic arthritis; PSO: psoriasis; Pt: patient; PVD: peripheral vascular disease; RIS: risankizumab; SEC: secukinumab; SPE: spesolimab; T2DM: Type 2 diabetes; TC: tinea cruris; TIL: tildrakizumab; UPA: upadacitinib;
USTE: Ustekinumab; y: years.
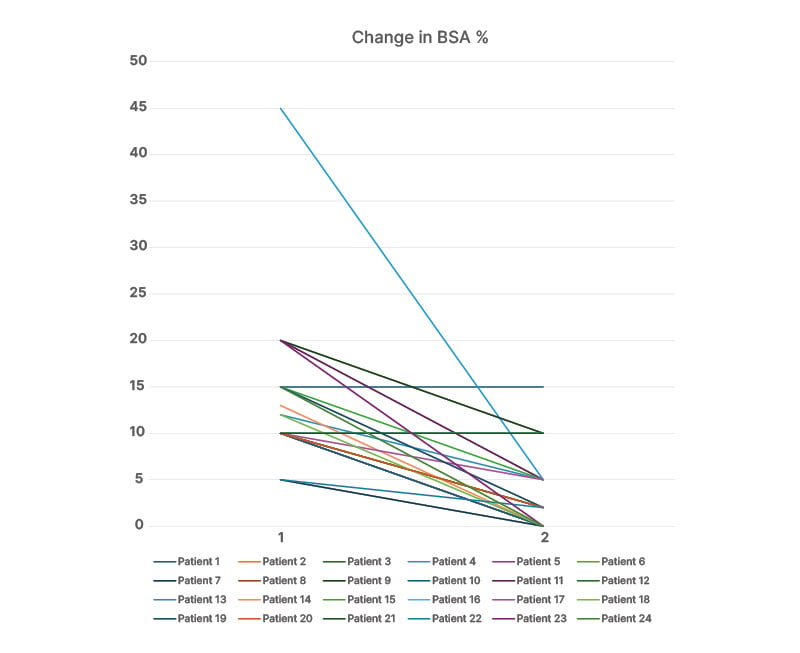
Figure 1: Change in body surface area (%) from initial follow-up visit.
Immediately prior to bimekizumab switch, 66.7% of patients were on an IL-17A or IL-17RA inhibitor, 20.8% of patients were on an IL-23 inhibitor, 8.33% of patients were on a TYK2 inhibitor, and one patient was on a TNF-α inhibitor. After a median of 184 days of therapy, BSA decreased to 3.8%. Overall, 83.3% of patients experienced a decrease in BSA after initiating bimekizumab, with 37.5% of patients obtaining a BSA of 0% (skin clear). In contrast, after an average of 119.25 days of therapy, 16.7% of patients experienced no change in disease severity. No patients clinically worsened while prescribed bimekizumab. Four patients experienced adverse events, with two of these resulting in bimekizumab discontinuation (gastrointestinal symptoms, injection site reaction [ISR]).
DISCUSSION
To the authors’ knowledge, this study is the first analysis of bimekizumab use in clinical practice in the United States. Aligning with clinical trials, this study reinforces bimekizumab as a useful therapeutic option for moderate-to-severe plaque psoriasis.1,2,5In the Phase III bimekizumab randomized clinical trials (BE READY, BE RADIANT, BE VIVID, BE SURE),1,5-7 patients were excluded if they had experienced a primary failure to an IL-17 inhibitor or had failed more than one biologic other than an IL-17 inhibitor. Further, in each of the four trials, less than half of the patients were bio-experienced.1,5-7 In contrast, 21 out of the 24 patients included in this study had attempted multiple biologics or systemic medications to treat their plaque psoriasis prior to attempting treatment with bimekizumab. Twenty-two patients had attempted an 1L-17 inhibitor, with 15 trialing secukinumab, 12 trialing ixekizumab, and eight trialing brodalumab (Table 1). Additionally, patients with certain comorbidities were excluded from the bimekizumab trials, for example those with hepatitis C infection. Although only one of the patients included in this study reported chronic hepatitis C infection, this patient had an improvement in affected BSA after starting therapy with no adverse events noted. Regarding special site involvement, nine patients had scalp psoriasis, with four achieving clearance, three achieving mild improvement, and two showing no improvement. Two patients had facial psoriasis that cleared after bimekizumab initiation, and three patients had palm/sole psoriasis with mild improvement. No patients presented with genital psoriasis.
Interestingly, oral candidiasis was a frequently reported adverse event in the bimekizumab clinical trials but was observed in only one patient in this study. This may be due to limited follow-up and small sample size. Also of note, ISR was reported in a small percentage in each of the four clinical trials; however, in this study, it was reported by two patients, with one patient requiring bimekizumab discontinuation due to ISR. As bimekizumab was only recently approved by the FDA, the duration of treatment with bimekizumab is limited by this constraint, as evident in this study.
CONCLUSION
Future real-world research investigating bimekizumab would benefit from increased size and longer follow-up. Despite limitations, this study supports bimekizumab as an appropriate option for treatment of moderate-to-severe plaque psoriasis among patients in real clinical practice, even those who previously failed multiple systemics and/or biologic therapies. Bimekizumab may be considered as a next-choice biologic therapy in biologic experienced patients who might have failed a prior biologic for psoriasis.






